Abstract
Introduction: Myelodysplastic Syndrome (MDS) and Chronic Myelomonocytic Leukemia (CMML) are clonal disorders driven by progressively acquired somatic mutations in hematopoietic stem cells (HSC) and characterized by the accumulation of blasts in the bone marrow and accompanying cytopenias. Hypomethylating agents (HMA) such as azacytidine (AZA) can modify the clinical course of MDS and CMML (Platzbecker et al Blood 2019). Although not curative, they are used as first-line therapies for high-risk patients who are ineligible or unable to access an allogeneic bone marrow transplant. Clinical improvement in response to HMAs is not accompanied by clearance or major shifts in mutant clones in the bone marrow (Unnikrishnan et al Cell Reports 2017, Merlevede et al Nature Communications 2016). However, it is unclear whether in patients with established MDS; (a) hematopoietic stem and progenitor cells with multiple mutations progress through to mature cells with comparable robustness to their counterparts with fewer or no mutations, or (b) the improvements in peripheral blood counts following HMA therapy is driven by residual wild-type hematopoietic stem and progenitor cells or by clones with particular combinations of mutations.
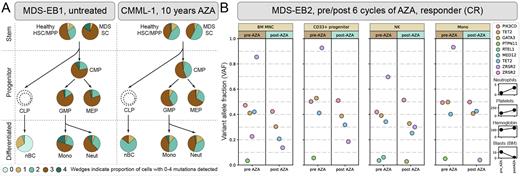
Methods: We index sorted hematopoietic stem (HSC, MPP) and progenitor cells (CMP, MEP and GMP) and mature blood cells (neutrophils (Neut), monocytes (Mono), and naïve B-cells (nBC): short lived cell types representative of current stem/progenitor output) from 3 MDS/CMML patients (1 treatment naïve, 2 treated >10 years with AZA) and performed targeted amplicon sequencing on thousands of single cells (n = 4248). In a second cohort (9 MDS patients; 6 responders, 3 non-responders) we sorted Mono, natural killer (NK) cells, and CD33+ progenitors and measured variant allele fraction (VAF) before and after 6 cycles of AZA.
Results: Single cell data showed the proportion of residual wild-type HSCs, and their contribution to mature myeloid cells, was minor (0 - 5.3% of HSCs, 0.3 - 4.5% of mature myeloid cells). Driver mutations were proportionately represented across multiple hematopoietic cell types, and even the most mutated stem and progenitor clones maintained their capacity to differentiate to mature myeloid and, in some cases, lymphoid cell types in vivo, irrespective of AZA treatment (Fig A). In a second cohort composed of paired pre- and post-AZA samples, clonal composition differed slightly between cell types; e.g. NK cells often harbored lower VAFs compared to monocytes and progenitors. We also observed treatment-associated reduction in a minority of mutated clones in this cohort. However, in all patients, clonal composition was remarkably similar before and after treatment, with highly mutated progenitors making a significant contribution to mature cells, even in patients showing reduced blast plus improved blood counts (Fig B).
Conclusions: Highly mutated immature cells contribute significantly to mature blood production in MDS and CMML, before and after AZA treatment. Our data highlight the key role of AZA therapy to promote output from mutant progenitors rather than simply eradicating them.
Disclosures
Mead:Sensyn: Consultancy, Speakers Bureau; Incyte: Consultancy, Speakers Bureau; Alethiomics Ltd: Consultancy, Current equity holder in private company, Other: Co-founder and equity holder, Research Funding; Pfizer: Consultancy, Speakers Bureau; Gilead: Consultancy, Speakers Bureau; Galecto: Consultancy, Research Funding, Speakers Bureau; AbbVie: Consultancy, Speakers Bureau; CTI: Consultancy, Speakers Bureau; Sierra Oncology: Consultancy, Speakers Bureau; Karyopharm: Consultancy, Speakers Bureau; Celgene/BMS: Consultancy, Research Funding, Speakers Bureau; Novartis: Consultancy, Honoraria, Research Funding, Speakers Bureau. Unnikrishnan:Celgene: Membership on an entity's Board of Directors or advisory committees, Research Funding. Hertzberg:Takeda: Consultancy, Honoraria; Roche: Consultancy, Honoraria; Otsuka: Consultancy, Honoraria; Novartis: Consultancy, Honoraria; Janssen: Consultancy, Research Funding; Gilead: Consultancy, Research Funding; Beigene: Consultancy, Honoraria. Polizzotto:AstraZeneca: Research Funding; BRII Biosciences: Research Funding; Celgene/BMS: Consultancy, Research Funding; CSL Behring: Research Funding; Eli Lilly: Research Funding; Emergent Biosciences: Research Funding; Gilead Pharmaceuticals: Consultancy, Research Funding; GlaxoSmithKline: Research Funding; Grifols: Research Funding; Janssen/J+J: Research Funding; Takeda: Research Funding. Papaemmanuil:Isabl Inc.: Current equity holder in private company, Current holder of stock options in a privately-held company, Other: CEO, Patents & Royalties: Whole genome cancer analysis; TenSixteen Bio: Current equity holder in private company. Pimanda:BMS: Research Funding; Abbvie: Honoraria, Membership on an entity's Board of Directors or advisory committees; Celgene: Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.